The Atom and Subatomic Particles (protons(+), neutrons( ), and electrons(-)
This is an awesome introductory interactive lesson to introduce students to the ATOM, including the structure of an atom and Atomic vocabulary
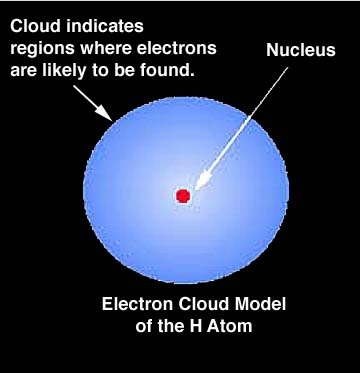
The basic atom
 Here is a Lithium atom
Here is a Lithium atom
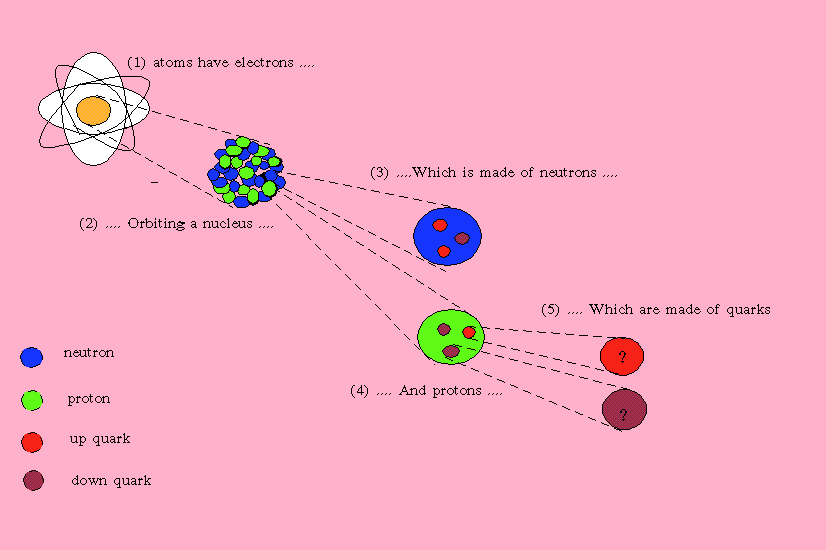
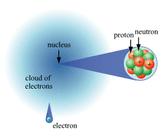
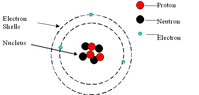
All matter is made up of atoms. Although we can go smaller than atoms, we will do that later. The basic atom consists of a nucleus surrounded by electrons going round the nucleus in orbit. Electrons are negatively charged.
The nucleus consists of:
The protons and neutrons have very nearly the same relative mass. The neutron has slightly more mass than the proton, but at this level we are going to say that the relative mass of both the proton and the neutron is 1.
The mass of a proton or neutron in kilograms is about 1.6 × 10-27 kg.
The mass of an electron is about 1/1800 the mass of a proton. The mass of an electron is about 9.1 × 10-31 kg.
Question: How many protons, neutrons, and electrons are there in this atom?
A: There are 3 protons, 4 neutrons, and 3 electrons in this lithium atom.
The nucleus consists of:
- Protons which are positively charged
- Neutrons that have no charge.
The protons and neutrons have very nearly the same relative mass. The neutron has slightly more mass than the proton, but at this level we are going to say that the relative mass of both the proton and the neutron is 1.
The mass of a proton or neutron in kilograms is about 1.6 × 10-27 kg.
The mass of an electron is about 1/1800 the mass of a proton. The mass of an electron is about 9.1 × 10-31 kg.
Question: How many protons, neutrons, and electrons are there in this atom?
A: There are 3 protons, 4 neutrons, and 3 electrons in this lithium atom.
Challenge Question: Explain how to find the # of neutrons in any atom
Subatomic Particles
Element Notation
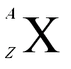
Elements are often written like this:
A is the total number of nucleons. Z is the total number of protons. |
| ||||||
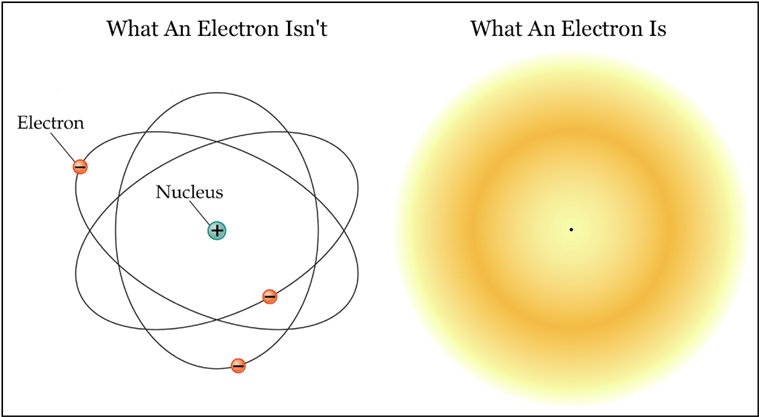
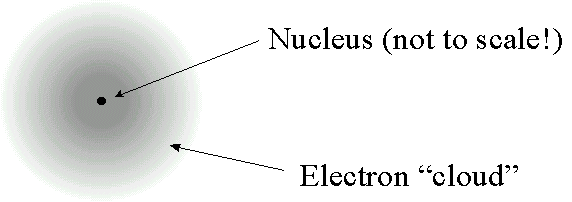
Modern atom models depict an electron shell or cloud
Challenge Question: How are models useful? How do we use models in everyday life?
Challenge Question: List two ways that electrons are different from neutrons.
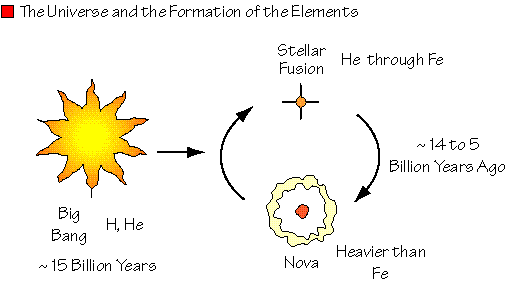
How elements are made in our Universe
Elements (about 114 right now known)
- An element is a substance made of only one type of atom.
- There are 92 naturally occurring elements found in the universe.
- Most atoms are cooked up in stars by nuclear fusion. Many of the heavy ones are fused in supernova explosions
- About 24 elements are not natural - people made them in particle accelerators
- A good way to think about all the different elements is to think of them as different flavors.
- You will need to learn the following elements symbols and their names: (Important) H, He, Li, Be, B, C, N, O, F, U, Na, Fe, Ar
States of Matter
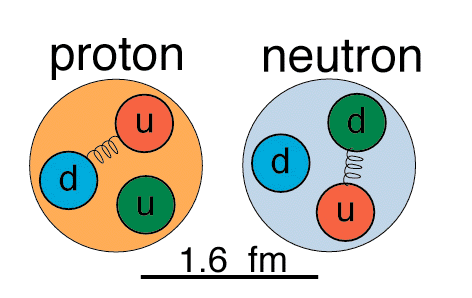
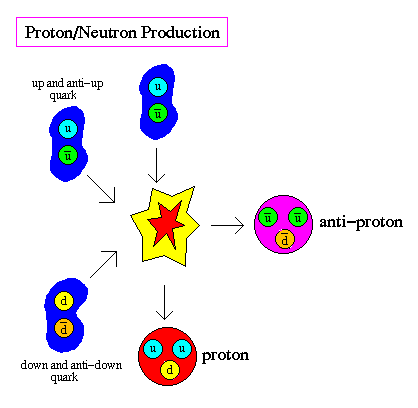
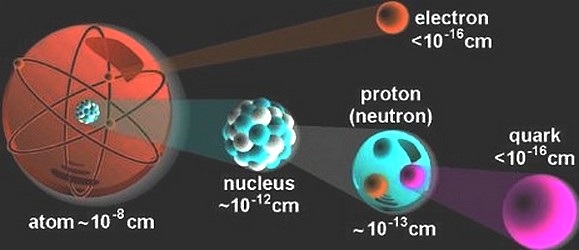
What in the world are quarks?

by domlane.
Explore more infographics like this one on the web's largest information design community - Visually.
Explore more infographics like this one on the web's largest information design community - Visually.
Large Hadron Collider
 Aerial view of Large Hadron Collider, with location of tunnel drawn on
Aerial view of Large Hadron Collider, with location of tunnel drawn on
Here is an infographic explaining what the Large Hadron Collider does.
http://greatprojectscampaign.com/large-hadron-collider.html
Here is the Science Channel's video on the device
http://www.sciencechannel.com/tv-shows/stuff-you-should-know/videos/stuff-you-should-know-large-hadron-collider.htm
http://greatprojectscampaign.com/large-hadron-collider.html
Here is the Science Channel's video on the device
http://www.sciencechannel.com/tv-shows/stuff-you-should-know/videos/stuff-you-should-know-large-hadron-collider.htm
E=mc2 not inside stars
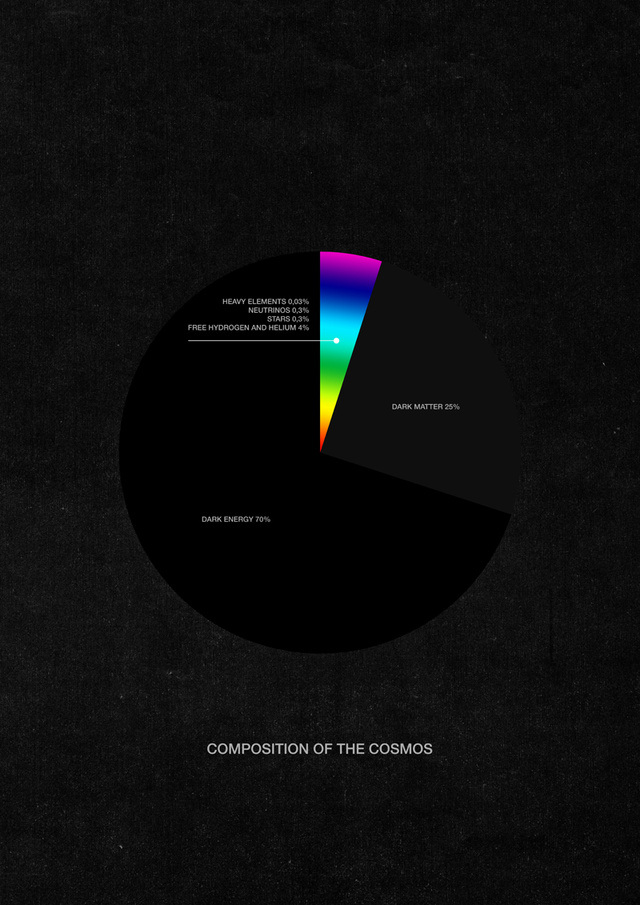
Neutrinos

Matter and the Atom Unit Essential Learning Targets
What you should Know, Understand and Be Able to Do after studying this unit
Learning Target 1: Students must understand that matter is classified based on its physical and chemical properties, and matter can undergo both physical and chemical changes
What you should Know, Understand and Be Able to Do after studying this unit
Learning Target 1: Students must understand that matter is classified based on its physical and chemical properties, and matter can undergo both physical and chemical changes
- NOTES: Classes and Properties of Matter
- PRACTICE: Classify these samples of matter
- PRACTICE: Online Matter Classification
- PRACTICE: Chemical/Physical Properties and Changes
- LAB: Chemical and Physical Properties
- PRACTICE: Flashcards for memorizing the names and symbols of the first 38 elements
- PRACTICE: Download the "Study Blue" app then click here to get interactive flashcards for memorizing the first 38 element names and symbols that you can use on your phone while you're in line at Starbucks!
- NOTES: Phases of Matter
- PRACTICE: Phases of Matter
- PRACTICE: Matter classes, properties, and phases Online Crossword Puzzle
- LAB: Separating Components of a Mixture by Distillation
- ONLINE QUIZ: Classes, Phases and Properties of Matter
- Atom Pre-test
- NOTES: Intro to the Atom - these notes describe the structure of the atom and properties of protons, neutrons, and electrons
- PRACTICE: Atom Chart - use your periodic table to fill in the missing information
- NOTES: Isotopes
- PRACTICE: aXz notation and atomic mass
- LAB: Beanium - determine the atomic mass of the fake element "Beanium"